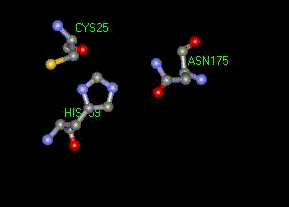
Figure 3. Papains' catalytic triad, consisting of Cys25, His159 and Asp175.
As most cysteine proteases, papain degrades protein as well as being an esterase. Papain has no primary specificity as to where it cleaves in proteins, although it does prefer P1=Arg or Lys. The active site of papain contains a catalytic triad consisting of Cys25, His159, and Asn175 (see figure 3), which is considered analogous to the Ser-His-Asp triad of the serine proteases.

Figure 3. Papains' catalytic triad, consisting of Cys25, His159 and Asp175.
The papain catalytic triad is deep in a cleft at the interface between the two domains (L and R). More specifically, Cys25 is part of the L1 alpha -helix at the surface of the domain, His159 is in a beta -sheet at the surface of the R-domain, while Asn175 is buried deep in the hydrophobic region of the enzyme (Vernet et al. 1995). The actual binding pocket can clearly be seen in a ball and stick model of papain (see figure 4) and in a model by secondary type with the catalytic triad in line form (see figure 5).
Figure 4. Papains' binding (as seen from the top of protein) pocket as seen in a ball and stick model.
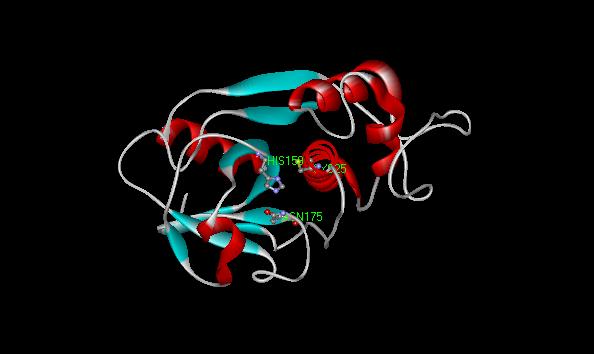
Figure 5. Papains' binding pocket (by secondary folding as seen from the top of protein) with the catalytic triad shown in line form.
Note how a substrate, such as a piece of protein, to be degraded would fit in the catalytic pocket in figure 6.
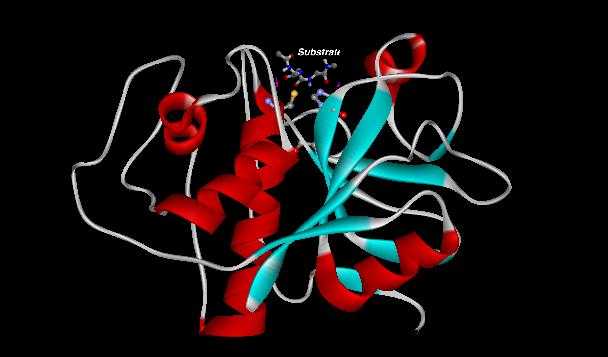
Figure 5. Binding of substrate into papains' catalytic pocket (substrate not to scale with regards to papain).
In catalytic pocket the active form the thiol group of Cys25 is deprotonated and it is stabilized by the positive charge of the imidazole ring of His159 (see figure 6). This in effect creates a stronger acid that is localized with in the catalytic pocket and establishes an environment that can cleave a peptide bond. Additionally the amide oxygen of the Asn175 hydrogen bonds to the N(epsilon2)H of His159, which further stabilizes the catalytic triad (Vernet et al. 1995). Drench et al. (1976) postulates that in the catalytic mechanism the imidazole moiety rotates over 30 A about the C(beta) -C(gama) (without disrupting its hydrogen bonding to Asn175), and the ring becomes equiplanar with Cys25-S(gama) and Asn175-O(delta1). Gln 19 can also be implicated in stabilizing the overall catalytic reaction.

Figure 6. The catalytic reaction of papains' active site, note the stabilization effects of His159 and Asp175 and the attack by the S of Cys25.
The catalyzed reaction can be represented schematically as a three-step process:
![]()
Free enzyme (E) and substrate (S) associate to for the Michaelis complex (E.S). Then the carbonyl carbon of the scissile ester or amide bond undergoes nucleophilic attack from the active site Cys25, which forms a covalent acyl-enzyme intermediate (E-A). The intermediate (E-A) later undergoes nucleophilic attack from a water molecule to yield the free enzyme and a carboxylic acid product (P2), the formation and decomposition of the acyl-enzyme intermediate are assumed to proceed through tetrahedral transition states (Wang et al. 1994).