Related Proteins
The two proteins of interest in the serpin family which are most closely related to ovalbumin are alpha-1-antitrypsin and anthrombin III.
Ovalbumin shows a primary sequence homology with 30 percent of alpha-1-antitrypsin. It was from studies of this protein that the reactive center for cleavage of ovalbumin has been determined (Figures 5 and 6). The structure of alpha-1-antitrypsin is shown in Figure 8.
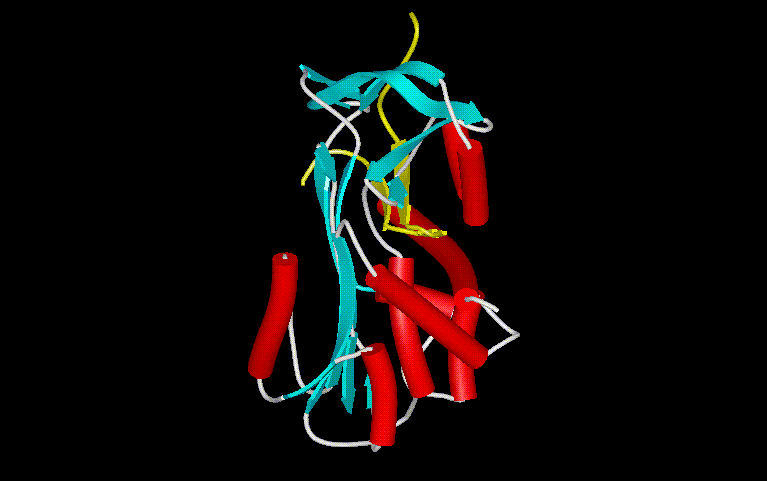 Figure 8. This structure is the relaxed form of alpha-1-antitrypsin. The native form is unknown, although it has been found to be similar with ovalbumin in active centers for cleavage. This protein has two domains present,Aand B. Domain B is highlighted in yellow, consisting only of antiparallel beta strands.
Figure 8. This structure is the relaxed form of alpha-1-antitrypsin. The native form is unknown, although it has been found to be similar with ovalbumin in active centers for cleavage. This protein has two domains present,Aand B. Domain B is highlighted in yellow, consisting only of antiparallel beta strands.
The other protein that has also been used in studying the function of ovalbumin is antithrombin III. The Arg-Ser bond has found to be similar in reactivity to ovalbumin's Ala-Ser bond. Also in this structure, antithrombin changes conformation into a relaxed state, in which ovalbumin continues to show contrary results of structural stability.
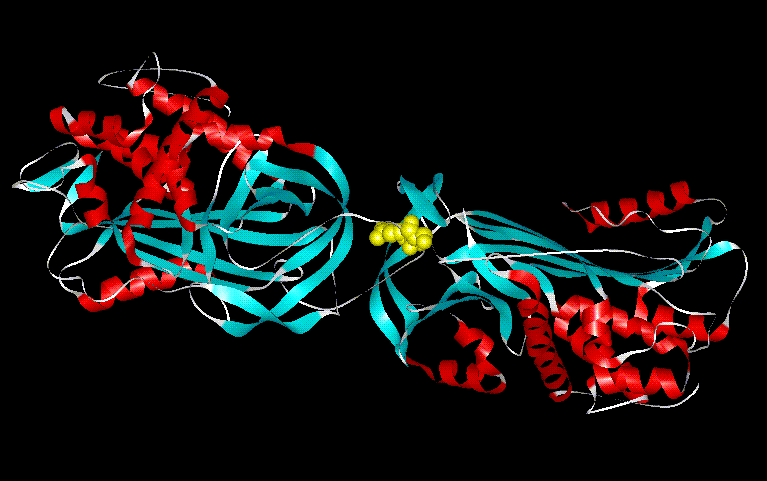
Figure 9. The protein, antithrombin consists of two domains, which are separated by the exposed loop in the center of the molecule shown above. Antithrombin's reactive center site is shown in this diagram as the highlighted region. This exposed loop results in cleavage of the molecule that similar to subtilisin's cleavage of ovalbumin.
These results have continuously shown up in research that relates to the study of ovalbumin. Methods used in determining this include H NMR readings as well as temperature comparisons. Upon proteolysis, antithrombin III and alpha-1-antitrypsin have shown significant increases in Tm. On the contrary, ovalbumin shows only a 4.4 degree drop in Tm upon proteolysis by subtilisin, with little alteration in structure or packing interactions.
 Figure 8. This structure is the relaxed form of alpha-1-antitrypsin. The native form is unknown, although it has been found to be similar with ovalbumin in active centers for cleavage. This protein has two domains present,Aand B. Domain B is highlighted in yellow, consisting only of antiparallel beta strands.
Figure 8. This structure is the relaxed form of alpha-1-antitrypsin. The native form is unknown, although it has been found to be similar with ovalbumin in active centers for cleavage. This protein has two domains present,Aand B. Domain B is highlighted in yellow, consisting only of antiparallel beta strands. 