Mechanism
The penicillin-binding-proteins (PBPs) are involved in the final stage of bacterial cell wall biosynthesis. The PBP-catalyzed hydrolysis leads to the formation of a serine-ester-linked acyl-enzyme intermediate. Marked by the D-Ala-D-Ala-carboxypeptidases, strands of peptidoglycan are hydrolyzed at the terminal D-Alanine residue. This initial step is accompanied by either the hydrolysis of the intermediate to generate a free D-Alanine terminus or transfer of the free amino terminus to another peptide chain, known as transpeptidation. Transpeptidation allows these acyl-enzyme intermediates to stabilize the bacterial cell wall against hypotonic lysis.
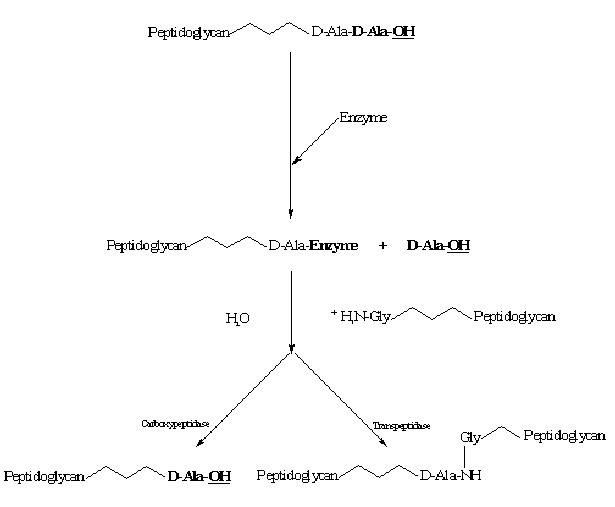 The process of bacterial cell wall synthesis. The strands of peptidoglycan are targeted by the D,D-carboxypeptidases, resulting in the hydrolysis of the terminal D-Alanine residue. The acyl-enzyme intermediate can then be deacylated by water or an amino acceptor, the latter resulting in a stabilized crosslinked structure (Richmond, 1994).
The process of bacterial cell wall synthesis. The strands of peptidoglycan are targeted by the D,D-carboxypeptidases, resulting in the hydrolysis of the terminal D-Alanine residue. The acyl-enzyme intermediate can then be deacylated by water or an amino acceptor, the latter resulting in a stabilized crosslinked structure (Richmond, 1994).
b-lactamases, undergoes a similar acylation by the b-lactam. However, unlike the previously mentioned penicillin-binding proteins, b-lactamases rapidly deacylate the penicilloyl complex, rendering b-lactam useless. The rate of deacylation of this acyl-enzyme intermediate resulting from interaction with b-lactam antibiotics with penicillin-binding-proteins is slow, therefore the bacterium is deprived of the biosynthetic function of these enzymes, an event that results in bacterial death.
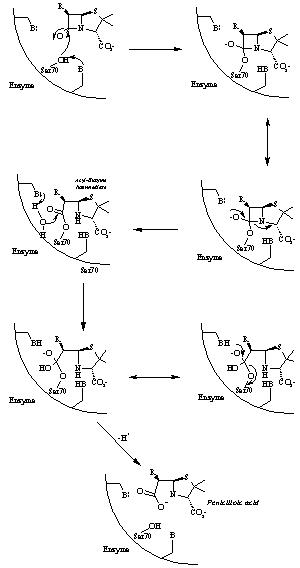
A mechanism for the hydrolysis of the b-lactam antibiotics by b-lactamase. The active site serine forms a acyl-enzyme intermediate with the antibiotics which is hydrolyzed by the transfer of a water molecule from Glu166 (Richmond, 1994)
On the basis of a similarity in the three-dimensional fold for class A and C b-lactamases with penicillin-binding-proteins, these proteins are believed to be related by a common ancestry (Bulyche, 1997). Even though the theory that the b-lactamases evolved from penicillin-binding-proteins was initially based on the substrate analogy, the penicillin-binding-proteins and the class A enzymes have also been shown to contain homologous sequences near the active site serine and have similar overall tertiary structure (Richmond, 1994).
The process of bacterial cell wall synthesis. The strands of peptidoglycan are targeted by the D,D-carboxypeptidases, resulting in the hydrolysis of the terminal D-Alanine residue. The acyl-enzyme intermediate can then be deacylated by water or an amino acceptor, the latter resulting in a stabilized crosslinked structure (Richmond, 1994).