
Actinidin is a globular protein with a structure very similar to that of papain (a previously isolated sulphydryl protease). To try and better understand the structure of Actinidin we will break it down into four main parts: primary structure, secondary structure, tertiary structure, and quaternary structure.
The primary structure of Actinidin in general can be experessed by its sequence linkages as shown in the following figure:

Fig 1. Actinidin Sequence Map
From this figure you can see the sequence of the 220 residues and their interactions. The structure's N-terminal end is composed of a Leu residue and its C-terminal end of a Tyr residue. There are seven Cys residues that form three disulphide bridges.
Actinidin contains three disulfide bridges that are located (as noted in Fig 1.) between residues 25 and 65, 56 and 98, & 156 and 206. For a better picture of where these bridges occur three dimensionsally on the protein please follow the link bellow.

Actinidin has been identified as having a double domain structure, with the active site located on the top surface between the two domains. The three dimensional shape of the protein is held together by hydrogen bonds, C and N-terminal "straps," and the previously mentioned disulphide bridges. Actinidin is categorized as an a + b protein. It contains alpha helix and beta sheets that are separated along the residue sequence. Domain one consists mainly of helical segments, while domain two is built around an irregular twisted beta sheet (Figure 2).

Figure 2. Domain Breakdown of Secondary Structure.
 Domain I, as mentioned above, has several pieces of alpha helix, but no significant beta sheet structure. Through the center of the molecule lies a five-turn alpha helix which makes up a large portion of the interdomain interface (residues 25-42). Another helix lines the top of the molecule beginning with a bifurcated hydrogen bond and donating side-chains to the domain core (residues 69-81). Both helicies are normal despite slight deviations from the ideal helical angles. With all carbonyl groups having a tendency to tilt outward from the helical core axis this seems appropriate. Domain I also contains two additional helical sections that are short and act very close to ideally (residues 50-57, 99-103). Each of the alpha helicies has a polar and non-polar side (including those in domain II). This fact allows the central helix of domain I, while mostly burried, to contribute its polar side chains to the polar interface between domains I and II.
Domain I, as mentioned above, has several pieces of alpha helix, but no significant beta sheet structure. Through the center of the molecule lies a five-turn alpha helix which makes up a large portion of the interdomain interface (residues 25-42). Another helix lines the top of the molecule beginning with a bifurcated hydrogen bond and donating side-chains to the domain core (residues 69-81). Both helicies are normal despite slight deviations from the ideal helical angles. With all carbonyl groups having a tendency to tilt outward from the helical core axis this seems appropriate. Domain I also contains two additional helical sections that are short and act very close to ideally (residues 50-57, 99-103). Each of the alpha helicies has a polar and non-polar side (including those in domain II). This fact allows the central helix of domain I, while mostly burried, to contribute its polar side chains to the polar interface between domains I and II.
 Domain II only contains two helical structures, a three-turn alpha helix (residues 120-129) and a two turn helix (residues 142-147). This helix lies on the outside of the moleculle running at a near perpendicular angle to the core five-turn helix of domain I. The rest of domain II is composed of an extensive, irregular beta sheet. The sheet is largely buried, contributing sidechains that constitute the make up of the core of domain II. The sheet consists of four antiparallel strands, including a classic b-bulge (residues 167-168, opposite residue 179). Actinidin's large beta sheet has a slight right handed twist which barring some residues (7, 167,168, 192) with non-standard angles behaves fairly commonly. In addition to the main beta sheet, domain II contains a short piece of antiparallel beta structure consisting of residues 111-116 and 212-216. The lack of interruptions in the hydrogen bonding pattern and the hydrogen bond lengths within these sheets are typical, showing little impact due to some water bonding partners.
Domain II only contains two helical structures, a three-turn alpha helix (residues 120-129) and a two turn helix (residues 142-147). This helix lies on the outside of the moleculle running at a near perpendicular angle to the core five-turn helix of domain I. The rest of domain II is composed of an extensive, irregular beta sheet. The sheet is largely buried, contributing sidechains that constitute the make up of the core of domain II. The sheet consists of four antiparallel strands, including a classic b-bulge (residues 167-168, opposite residue 179). Actinidin's large beta sheet has a slight right handed twist which barring some residues (7, 167,168, 192) with non-standard angles behaves fairly commonly. In addition to the main beta sheet, domain II contains a short piece of antiparallel beta structure consisting of residues 111-116 and 212-216. The lack of interruptions in the hydrogen bonding pattern and the hydrogen bond lengths within these sheets are typical, showing little impact due to some water bonding partners.
There are twelve turns within the molecule. Three within domain I and nine in domain II. These turns connect segments of the beta sheet, connect helicies together, or connect the beta sheet to a helix. I have included a piece of the structure here that does not seem to fit the category of alpha helix or beta sheet, but forms some sort of "S" shaped piece of two interlocking turns (residues 201-210). Other than this piece, the basic structure of the turns reflects normal hydrogen bonding patterns (either with other structural pieces or water molecules).
The Hydrogen Bonds serve two main purposes (outside of the active site). These bonds impact the secondary and tertiary shape that the structural elements take. Actinidin's residues form Hydrogen Bonds within the protein as well as with solvent molecules. These interactions play a large role in protein folding a overall three dimensional shape.
The cores of both domains are dominantly buried non-polar side-chains. There are however, some non-polar side-chains that are exposed to the external environment. Some of these residues form a hydrophobic "patch" on the surface of the protein (Ile151, Val175, Leu197, Val200 and Thr153), that has no identified function. Residues Phe76, Tyr147, and Phe152 have one edge of their aromatic rings within the core and the other edge accessible to the solution. The aromatic side-chains on the surface roughly pack in an "edge-on" manner. The partially exposed rings exhibit no p-p interactions as shown by the fact that they do not pack plane-to-plane.
Acidic and basic side chains are evenly distributed about the surface of the protein since at pH 6 they are usually charged and available to interact with solvent molecules. There are more acidic side-chains than basic, which is reflected in the low isoelectric point (pI ~3.1), but there are no obvious pockets of charge concentration. There are four internal charged polar groups that do not yet have an identified functional role. However, there are some charged polar groups located in the domain interface, but these chains are located where they can neutralize each other while forming hydrogen bonds with the internal water molecules.
The side-chains of Trp and Tyr are all involved in hydrogen bonding. Some project into solution forming hydrogen bonds with water molecules and others project into the molecule forming intramolecular hydrogen bonds. Several of the Tyr residues are crutial in internal hydrogen bonding for the stability of loosely bound loops located on the exterior of the molecule or near the beta sheet of domain II.
There are only three places where the polypeptide chain crosses between the two domains (residues 16-22 on one side and 115-119 & 212-214 on the other). The interior of this interface is largely polar with only three non-polar residues (Ala32, Val165, Pro132). However, the polar interface is encircled by nonpolar residues which seem to act as a shield, protecting the interior of the interface from the external environment. Probably the most unique aspect of the domain interface are the internal water molecules. This structural element is likely the result of the water molecules becoming trapped during folding. It seems likely by this model that the two domains folded up independently (each with a polar exterior) and then joined together. While apparently random, this structural feature seems to have functional importance. It has been suggested by E.N. Baker (1980) that these internal water molecules allow the protein more flexibility, due to an elastic "breathing" capability.
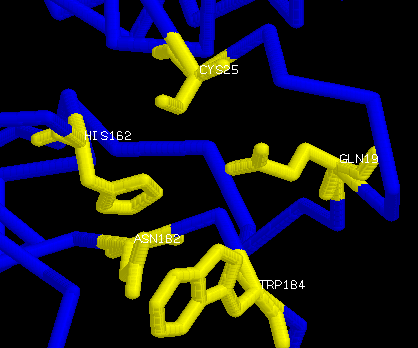 The two key groups for catalysis are a sulphydryl group and an imidazole group. The arrangement of Cys25, with its free sulphydryl group, and of His162, the only histidine in actinidin, along with some other residues in close proximity act as the active site of the molecule. The mechanism for the activity of the protein closely mimics that of papain. Cys25 and His162 are the residues the main. The orientation of Gln19 places a NH2 in the active site where it could interact with a substate carbonyl oxygen. The main-chain carbonyl groups of residues 66, 68, and 161 and the main-chain NH groups of residues 25 and 68 also point toward the active site providing units for substrate interaction. The side-chain of Met211 stretches accross the end of the substrate binding pocket making it noticably shorter than in papain. The other binding sites are located around the mouth of the active-site cleft (some in and some out) and near Met211. The reaction that occurs in the active site is as such:
The two key groups for catalysis are a sulphydryl group and an imidazole group. The arrangement of Cys25, with its free sulphydryl group, and of His162, the only histidine in actinidin, along with some other residues in close proximity act as the active site of the molecule. The mechanism for the activity of the protein closely mimics that of papain. Cys25 and His162 are the residues the main. The orientation of Gln19 places a NH2 in the active site where it could interact with a substate carbonyl oxygen. The main-chain carbonyl groups of residues 66, 68, and 161 and the main-chain NH groups of residues 25 and 68 also point toward the active site providing units for substrate interaction. The side-chain of Met211 stretches accross the end of the substrate binding pocket making it noticably shorter than in papain. The other binding sites are located around the mouth of the active-site cleft (some in and some out) and near Met211. The reaction that occurs in the active site is as such:


Actinidin is a globular protein that very closely resmbles the three dimentional structure and folding pattern of papain. The molecule is generally polar on its surface to allow for interaction with water molecules. These surface residues commonly have charged polar groups. The core of the protein is generally nonpolar resulting from the grouping of hydrophobic interactions. The uncharged polar groups are commonly buried hydrogen bond donors and acceptors. These three types of sidechains play a role in the folding of the molecule, aiding in giving it the three dimensional structure that is observed.
 There is no quaternary structure in the Actinidin since it is a single chain of residues. However, we can look briefly at the crystal packing structure of the molecule that was isolated for use in analysis (picture taken from E.N. Baker [1980]). Interactions between molecules in the crystal structure take three main forms: hydrogen bonds, van der Waals' effects, and water bridges. There are three main types of interaction sites that can be found in the crystal structure. A regions (not shown) which have several hydrogen bonds, van der Waals' effects, and water bridges. B regions have few van der Waals' effects, but similar amounts of hydrogen bonds and water bridges. C regions have no van der Waals' effects, and similar amounts of hydrogen bonds and water bridges.
There is no quaternary structure in the Actinidin since it is a single chain of residues. However, we can look briefly at the crystal packing structure of the molecule that was isolated for use in analysis (picture taken from E.N. Baker [1980]). Interactions between molecules in the crystal structure take three main forms: hydrogen bonds, van der Waals' effects, and water bridges. There are three main types of interaction sites that can be found in the crystal structure. A regions (not shown) which have several hydrogen bonds, van der Waals' effects, and water bridges. B regions have few van der Waals' effects, but similar amounts of hydrogen bonds and water bridges. C regions have no van der Waals' effects, and similar amounts of hydrogen bonds and water bridges.